Abstract
INTRODUCTION
Cytokines are involved in many processes, including hematopoiesis and inflammation. Aging is associated with the onset of clonal hematopoiesis (CH) of indeterminate potential, putatively associated with a higher risk of progression to hematological malignancies such as myelodysplastic syndromes or acute myeloid leukemia. Moreover, CH may participate to create a pro-inflammatory environment contributing to the pathogenesis of age-related diseases, such as cardiovascular diseases. This is likely to be driven by or translated in changes in bone marrow (BM) and/or peripheral blood (PB) soluble factors for which reference values still remain unclear, because BM cytokines levels have never been determined in strictly selected healthy people. Indeed, control BM samples classically used in studies are from subjects undergoing surgeries for non-hematologic causes, such as total hip replacement or cardiac surgery, patients suffering from immune thrombocytopenic purpura, brain death patients or allogeneic BM donors. In this study, the BM and PB plasma concentrations of 49 hematopoietic and inflammatory cytokines were measured in a representative panel of 94 healthy adult volunteers and the results were analyzed considering their age and presence of CH.
METHOD
Ninety-four healthy donors aged from 18.6 to 80.1 years old (yo), including 58 women were recruited for this study (HEALTHOX protocol, CPP Tours, AFSSAPS identifier ID-RCB: 2016-A00571-50 and ClinicalTrials.gov # NCT02789839). The presence or absence of CH (>1% of variant allele frequency) in this cohort is already known (Guermouche H, Ravalet N et al, Blood Adv 2020;4(15):3550-3557). BM samples were obtained through sternal aspiration using a classical procedure in France, and PB sampling was performed at the same time by venipuncture. Samples were collected on sodium heparin or EDTA, centrifuged twice (1200 g, 10 min, 20 C°), aliquoted and stored at -80°C. A 48-plex human cytokine panel assay was used to quantify 48 human cytokines in PB and BM plasmas [beta-NGF, CCL2, CCL3, CCL4, CCL5, CCL7, CCL11, CCL27, CLEC11A, CXCL1, CXCL8, CXCL9, CXCL10, CXCL12, FGF-2, G-CSF, GM-CSF, HGF, IFN-alpha-2, IFN-gamma, IL-1 alpha, IL-1 beta, IL-1ra, IL-2, IL-2-RA, IL-3, IL-4, IL-5, IL-6, IL-7, IL-9, IL-10, IL-12, IL-12B, IL-13, IL-15, IL-16, IL-17A, IL-18, KITLG, LIF, LT-alpha, M-CSF, MIF, PDGF subunit B, TNF, TNFSF10, VEGF-A]. MIF and FLT3L quantification were performed by ELISA. Regarding CH, the controls were subjects older than 50 yo without CH. Statistical analyses were performed with the R (3.6.3) and Rstudio version 1.2.5042 (www.rstudio.org) software. Comparisons were computed by Wilcoxon and Kruskal-Wallis tests. All pairwise multiple comparisons were performed using Dunn's-test for multiple comparisons of independent samples (PMCMR package). Correlations between cytokine levels in PB and BM were tested with Pearson and Spearman methods. Correlation matrices were plotted using the "corrplot" package.
RESULTS
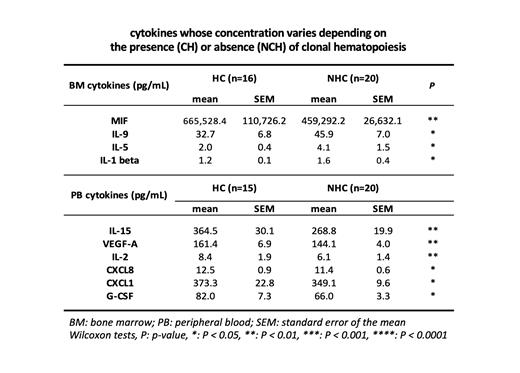
CH was detected in 16 volunteers, mostly in individuals over 50 yo. BM and PB plasma samples were studied in 3 age-groups: 18-40, 40-60 and 60-80 yo. With aging, variations were observed for 18 BM cytokine levels, with 7 increasing (FLT3L, CXCL9, HGF, FGF-2, CCL27, IL-16, IL-18) and 8 decreasing (G-CSF, TNF, IL-2, IL-15, IL-17a, IL-4, LT-alpha, IL-1 alpha). In PB, 10 cytokines significantly increased with age (CXCL9, FLT3L, CCL27, CXCL10, HGF, CCL11, IL-16, IL-6, IL-1 beta, CCL2). CH was associated with significantly higher BM levels of MIF and IL-1 beta, lower BM levels of IL-9 and IL-5 and higher PB levels of IL-15, VEGF-A, IL-2, CXCL8, CXCL1 and G-CSF (Table).
CONCLUSION
In this study and for the first time, we concomitantly analyzed BM and PB concentrations of a panel of hematopoietic and inflammatory cytokines in a cohort of strictly selected healthy volunteers. In addition to the establishment of reference values, useful for various biological studies, and correlations between blood and BM levels, we identify variations in the BM of key cytokines according to age and CH. The differences in these cytokine concentrations, either as causes or as consequences, may shape both the BM microenvironment and hematopoietic processes, eventually leading to the beginning of age-related myeloid malignancies or inflammatory conditions.
Hirsch: Daiichi Sankyo Oncology: Consultancy; Novartis Pharma: Consultancy. Suner: Sanofi - Genzyme: Consultancy. Delhommeau: Celgene: Consultancy; BMS: Consultancy; Novartis: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal